Hypertriglyceridemia is an independent risk factor
for only colorectal tubular
adenoma in male
Masafumi
Tabuchi 1) 2), Joji Kitayama 2) Kazushige Kawai 2), Kenji
Hatano and 2) and Hirokazu Nagawa2)
1)
Nakameguro Digestive Disease
Clinic
2)
Department of Surgical Oncology, University of Tokyo
Introduction
Many
studies have shown the positive correlation between lipid especially
cholesterol and the incidence of colorectal adenoma or cancer. Most of these results suggests that high
intake of dietary fat tends to promote colon
carcinogenesis 1,
2
3. The fecal excretion of
cholesterol was elevated in patients with colon cancer and adenomatous polyps,
suggesting a possibility that the direct contact of cholesterol or its
derivatives on colonic epithelia may be important for colonic carcinogenesis 4 On the serum lipid level, early studies showed an inverse association between
cholesterol level and colon cancer, while contradictory results have also been
reported 5, 6 However, another study has shown the
small but significant positive association between serum cholesterol levels and
the risk of colorectal adenomas 7 In addition, positive correlation with
colonic adenomas with atherosclerotic diseases has been reported 8. Those facts suggest that low serum
cholesterol associated with malignancies may be a consequence of the impaired
nutritional status rather than a cause of cancer.
As
compared with cholesterol, there has been less information on the relationship
between serum triglyceride and colorectal neoplasm. The striking similarity in
lifestyle risk factors for colorectal cancer and insulin resistance, suggested that the
hypertriglyceridemia as well
as hyperinsulinemia and hyperglycemia
associated with insulin resistance can support the development of colon cancer 9-12 Animal
study also indicates that development of aberrant crypt foci is associated with
circulating triglyceride as well as glucose, suggesting a possible role of
triglyceride on colon cancer development 13. On the contrary, other reports failed to detect significant association between serum levels of triglyceride
as well as cholesterol
and risk
of colorectal cancer in human 14, 15 and animal studies16 .
Thus, the contribution of circulating lipid level on
colonic neoplasm development has not been satisfactorily determined. In our clinic, we have performed large
number of total colonoscopy for the screening of colon cancer and intensively
tried to find the colonic adenomatous polyps, since it is generally
accepted that most colorectal cancers arise from adenomatous polyps. From the
experience, we retrospectively analyzed the relationship between serum level of
cholesterol or triglycerides and the presence of colonic adenoma.
Methods and Patients
A
total of 4887 patients underwent total colonoscopy more than three times for
the screening of colon cancer and precancerous lesions from 1995 to 2003. Total colon was carefully examined with
high resolution magnified chromo-endoscope. More
than 20
minutes were spent for observation in
each colonoscopy in order to
avoid the failure to detect
small lesions. All the
adenomatous polyps
including mucosal cancers detected
in colonoscopy were
simultaneously resected by endoscopic polypectomy technique and assessed for
pathological examination. A total
of 110 invasive cancers were found and surgically resected during the periods.
The fasten serum level of two major lipids, TC (Total cholesterol), Low density lipoprotein (LDL),
and/or Triglycerides (TG), were measured before the colonoscopy. The data for TC and TG
were available in 4883 and 4874
cases, respectively. Then, the
relationship between the incidence of colonic neoplasm and fasten serum levels
of these lipids was retrospectively evaluated. The statistical analysis was performed
with JUMP software. The serum lipid
levels in patients were analyzed with ANOVA and Fisher’s exact test. The odds ratios were evaluated with
multiple logistic regression tests.
P values less than 0.05 were determined as statistically significant.
Results
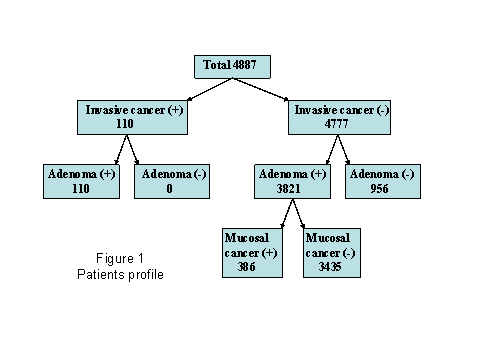
In
our careful endoscopic observation, invasive cancer was found in 110 (2.3%) in
4887 patients. All the patients
also had the 1-77 adenomatous polyps in other part of the colon. In the remaining 4777 patients, no
adenomas were found only in 956 (19.6%) patients, whereas at least one adenoma
can be detected in other 3821 (78.2%) patients, and all of them were
endoscopically removed simultaneously.
In these 3931 patients with adenomas (including patients with invasive
cancer), only one adenoma was detected in 660 (13.5%) patients, 2 and 3 adenomas
were found in 549 (11.2%) and 458 (9.4%) patients, respectively. In other patients, 1934 (39.6%) patients
had from 4 to 9 while other 788 (16.1%) patients had more than 10 adenomas in
total colon. Pathological
examinations showed that mucosal cancer were observed in 386 (9.9%)
patients. Most of the resected
adenomas, whether associated with cancer or not, were histologically diagnosed
as tubular adenoma, and thus tubular adenoma was detected in 3928 (80%) cases
in total patients. Villous adenoma,
and serrated adenoma were observed in 53 (1.4%) and 36 (0.9%) cases,
respectively (Fig.1).
At
the first step, we analyzed if the serum levels of TC and TG of patients with cancer
or adenoma were different from those in patients without these lesions (Fig.2).
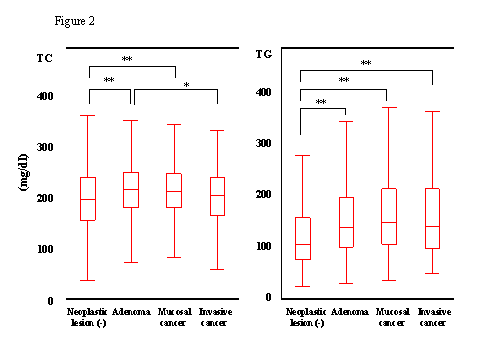
As examined with ANOVA, both values were significantly elevated in patients
with adenomas as compared with patients without any neoplastic lesions. (TC 207.6±29.5 vs 199.5±34.3, p<0.001; TG 135.0±82.2 vs 108.7±71.5 P<0.001).
Patients with invasive or mucosal cancer showed the same levels of TG
(142.0±75.1,
139.3±91.6, respectively) as those with adenomas that were
significantly higher than that in patients without adenomas as well as cancers
(p<0.001). In TC, however,
patients with mucosal cancer also showed same level as those with adenomas
(206.2±28.7),
while patients with invasive cancer showed relatively reduced level (200.8±28.3), that was not significantly different from that
in patients without neoplastic lesions.
LDL cholesterol was examined in 1483 patients and showed the similar
profile as TC .
Next, we examined the pathological component of these differences of lipid levels in patients with adenomas.
|
Table 1. Serum levels of Total
cholesterol (TC) and Triglyceride (TG) in patients |
|
|
|
|
|
|
|
|
with tubulatr, villous and serrated
adenomas |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Pathological finding |
TC |
n |
p value |
|
TG |
n |
p value |
|
tubular adenoma |
|
|
|
|
|
|
|
|
abesnt |
199.6±34.4 |
958 |
|
|
108.9±71.0 |
954 |
|
|
present |
207.3±29.3 |
3925 |
p<0.0001 |
|
135.8±81.8 |
3920 |
p<0.0001 |
|
villous adenoma |
|
|
|
|
|
|
|
|
abesnt |
205.8±30.6 |
4831 |
|
|
130.5±80.7 |
4821 |
|
|
present |
211.5±24.6 |
52 |
N.S |
|
132.0±60.8 |
53 |
N.S |
|
serrated adenoma |
|
|
|
|
|
|
|
|
abesnt |
205.8±30.6 |
4848 |
|
|
130.4±80.3 |
4838 |
|
|
present |
208.8±24.1 |
35 |
N.S |
|
149.3±102.3 |
36 |
N.S |
As shown in Table 1, the serum levels of these lipids were significantly different
in patients with tubular adenomas, whereas not in patients with villous or
serrated adenomas. This indicates
that hyperlipidemia has the specific association with the development of
tubular adenoma, but not the development of carcinomas as well as other types
of adenomas.
Therefore, we evaluated the association between serum lipid levels and tubular adenomas. Since TC and TG are known to be closely related with age and gender, we analysed the odds ratios adjusted with these factors.
|
|
Table 2. Univariate analysis of risk for
tubular adenoma |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Tubular adenoma |
|
|
negative (%) |
|
positive (%) |
|
p value |
|
|
|
total |
|
|
|
|
|
|
Age |
|
|
|
|
|
|
|
|
|
10-56 |
2475 |
765 (30.9) |
|
1710 (69.1) |
|
|
|
|
57-94 |
2412 |
194 (8.0) |
|
2218 (92.0) |
|
<0.0001 |
|
Gender |
|
|
|
|
|
|
|
|
|
male |
2997 |
402 (13.4) |
|
2595 (86.6) |
|
|
|
|
female |
1890 |
557 (29.5) |
|
1333 (70.5) |
|
<0.0001 |
|
Hypercholesterolemia |
|
|
|
|
|
|
|
|
|
Negative: <220mg/dl |
3318 |
695 (21.0) |
|
2623 (79.0) |
|
|
|
|
Positive: =>220mg/dl |
1565 |
263 (16.8) |
|
1302 (83.2) |
|
<0.0001 |
|
Hypertriglyceridemia |
|
|
|
|
|
|
|
|
|
Negative: <150mg/dl |
3488 |
784 (22.5) |
|
2704 (77.5) |
|
|
|
|
Positive: =>150mg/dl |
1386 |
170 (12.3) |
|
1216 (87.7) |
|
<0.0001 |
As shown in Table 2, univariate analysis showed that male and old patients had colonic adenomas more frequently than their counterparts. Then, we separately calculated the odds ratios for the change of 1standard deviation (SD) adjusted with age in male and female patients (Table 3).
|
Table 3. Adjusted Odds ratios and 95%CIs for
tubular adenoma by 1SD change |
|
|
|
|
|
|
|
|
in male or female patients |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total Cholesterol |
|
|
|
|
Triglyceride |
|
|
Total (3787) |
|
|
|
|
|
|
|
|
Gross OR |
1.3 |
(1.21-1.39) |
<0.0001 |
|
1.64 |
(1.48-1.82) |
<0.0001 |
|
Age adjusted OR |
1.01 |
(0.93-1.09) |
0.79 |
|
1.34 |
(1.21-1.48) |
<0.0001 |
|
Age, Gender adjusted OR |
1.07 |
(0.99-1.17) |
0.086 |
|
1.15 |
(1.04-1.26 |
0.0053 |
|
Age, Gender and TC or TG adjusted OR |
1.04 |
(0.96-1.13) |
0.334 |
|
1.13 |
(1.01-1.09) |
0.013 |
|
|
|
|
|
|
|
|
|
|
Male (2997) |
|
|
|
|
|
|
|
|
Gross OR |
1.32 |
(1.19-1.47) |
<0.0001 |
|
1.35 |
(1.19-1.56) |
<0.0001 |
|
Age adjusted OR |
1.09 |
(0.98-1.23) |
0.13 |
|
1.24 |
(1.09-1.43) |
0.0015 |
|
Age and TC or TG adjusted OR |
1.04 |
(0.91-1.07) |
0.555 |
|
1.23 |
(1.07-1.42) |
0.0041 |
|
|
|
|
|
|
|
|
|
|
Female (1890) |
|
|
|
|
|
|
|
|
Gross OR |
1.49 |
(1.34-1.65) |
<0.0001 |
|
1.42 |
(1.25-1.64) |
<0.0001 |
|
Age adjusted OR |
1.08 |
(0.96-1.21) |
0.21 |
|
1.04 |
(0.92-1.17) |
0.56 |
|
Age and TC or TG adjusted OR |
1.06 |
(0.95-1.02) |
0.29 |
|
1.02 |
(0.91-1.16) |
0.7 |
Multiple logistic regression analysis showed the clear contrast between TC
and TG. When age and gender were
considered, TC lost the relationship with adenoma risk for either male or
female subpopulations. On the
contrary, TG was remained as a candidate of an independent risk factor for
tubular adenoma in total (p<0.05) and male (p<0.01) patients, but not in
female patients.
Finally,
we evaluated the relationship between the serum lipid level and the number of
tubular adenoma (Fig 3).
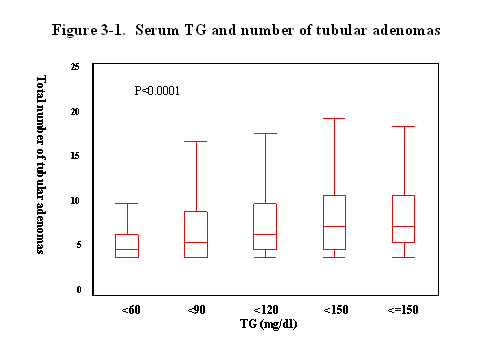
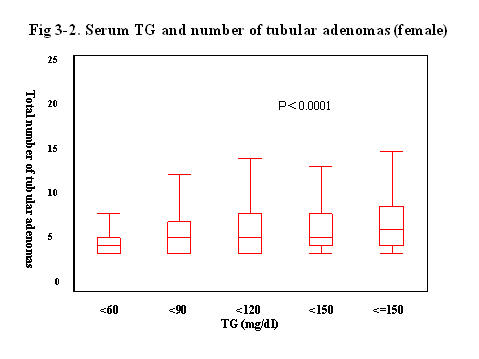
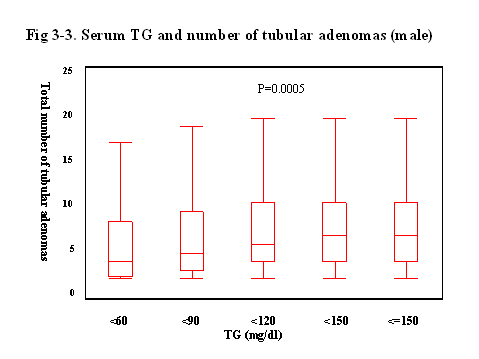
Interestingly, when the serum levels of TG was elevated, the average of the
number of tubular adenoma were gradually increased, and the correlation was statistically
significant (p<0.0001).
Conclusions and Discussions
Our
study clearly indicates that hyperlipidemia is frequently associated with
colonic adenoma and carcinoma.
These facts were often reported in previous papers. In this
retrospective large scale study, some new interesting facts were discovered
additionally.
1) Total cholesterol level was not a specific
risk factor for colonic tumors from multivariate analysis, possibly because of
the strong association with age.
2) Serum triglyceride level is remained as a
candidate of an independent risk factor for colonic tubular adenoma even after
multivariate analysis.
3) And moreover there was a gender difference
in the theory 2), which was observed only in male but not in female patients.
4) Villous tumor is not associated with serum
lipid level.
5) Serrated tumor is not associated with serum
lipid level neither.
6) Total cholesterol level in patients with
invasive cancer was rather decreased to the same level as patients without
adenoma, while TG levels in patients with invasive cancer kept at the same
level as those in patients with adenoma.
Serum cholesterol was not so linked to colonic tumor as serum triglyceride.
The link point of serum TG to colorectal tumor genesis was that of tubular
adenoma genesis. And the feature of this linkage was sex dependent. These facts
remind us of PPAR-γ, which diminishes
both tumor genesis and serum TG level, and interrupted by estrogen. PPAR-γmight be playing an important role in colorectal tubular adenoma genesis.
To elucidate this speculation, further study will be needed.
Previous studies have shown the same association between cholesterol level
and colonic villous tumors7. But the other paper17 showed the inverse
result. In the later paper the number of tumors was so small compared with our
study and the odds were just on the threshold. So we think that there is no
relationship between villous component and serum lipid level.
About relationship between serum lipids and serrated histological
component, there were no previous reports as far as we researched.
References
1.
Reddy BS. Dietary fat and its relationship to large bowel cancer. Cancer Res 1981;41(9 Pt 2):3700-5.
Available from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=6266660.
2.
Vogel VG, McPherson RS. Dietary epidemiology of colon cancer. Hematol Oncol Clin North Am
1989;3(1):35-63. Available from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=2537285.
3.
Potter JD. Risk factors for colon neoplasia--epidemiology and biology. Eur J Cancer 1995;31A(7-8):1033-8.
Available from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=7576987.
4.
Reddy BS, Wynder EL. Metabolic epidemiology of colon cancer. Fecal bile acids
and neutral sterols in colon cancer patients and patients with adenomatous
polyps. Cancer 1977;39(6):2533-9.
Available from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=872053.
5.
Neugut AI, Johnsen CM, Fink DJ. Serum cholesterol levels in adenomatous polyps
and cancer of the colon. A case-control study. Jama 1986;255(3):365-7. Available from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=3941515.
6.
Stemmermann GN, Heilbrun LK, Nomura AM. Association of diet and other factors
with adenomatous polyps of the large bowel: a prospective autopsy study. Am J Clin Nutr 1988;47(2):312-7.
Available from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=3341261.
7.
Mannes GA, Maier A, Thieme C, Wiebecke B, Paumgartner G. Relation between the
frequency of colorectal adenoma and the serum cholesterol level. N Engl J Med 1986;315(26):1634-8.
Available from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=3785334.
8.
Correa P, Strong JP, Johnson WD, Pizzolato P, Haenszel W. Atherosclerosis and
polyps of the colon. Quantification of precursors of coronary heart disease and
colon cancer. J Chronic Dis
1982;35(5):313-20. Available from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=7068807.
9.
McKeown-Eyssen G. Epidemiology of colorectal cancer revisited: are serum
triglycerides and/or plasma glucose associated with risk? Cancer Epidemiol Biomarkers Prev 1994;3(8):687-95. Available from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=7881343.
10.
Giovannucci E. Insulin and colon cancer. Cancer
Causes Control 1995;6(2):164-79. Available from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=7749056.
11.
Bruce WR, Wolever TM, Giacca A. Mechanisms linking diet and colorectal cancer:
the possible role of insulin resistance. Nutr
Cancer 2000;37(1):19-26. Available from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=10965515.
12.
Yamada K, Araki S, Tamura M, Sakai I, Takahashi Y, Kashihara H, et al. Relation
of serum total cholesterol, serum triglycerides and fasting plasma glucose to
colorectal carcinoma in situ. Int J
Epidemiol 1998;27(5):794-8. Available from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=9839735.
13.
Koohestani N, Chia MC, Pham NA, Tran TT, Minkin S, Wolever TM, et al. Aberrant
crypt focus promotion and glucose intolerance: correlation in the rat across
diets differing in fat, n-3 fatty acids and energy. Carcinogenesis 1998;19(9):1679-84. Available from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=9771941.
14.
Saydah SH, Platz EA, Rifai N, Pollak MN, Brancati FL, Helzlsouer KJ.
Association of markers of insulin and glucose control with subsequent
colorectal cancer risk. Cancer Epidemiol
Biomarkers Prev 2003;12(5):412-8. Available from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=12750235.
15.
Gaard M, Tretli S, Urdal P. Blood lipid and lipoprotein levels and the risk of
cancer of the colon and rectum. A prospective study of 62,173 Norwegian men and
women. Scand J Gastroenterol
1997;32(2):162-8. Available from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=9051877.
16.
Barton TP, Cruse JP, Lewin MR. Changes in serum lipids related to the presence
of experimental colon cancer. Br J Cancer
1987;56(4):451-4. Available from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=3689662.
17.
Houghton J, Lardieri GG, Zauber NP, Kim KH, Cable G. Effect of cholesterol
levels on villous histology in colonic adenomas. Dig Dis Sci 2000;45(5):896-9. Available from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=10795751.